Imagine the delicious smell of a family’s favorite home-cooked recipe passed down through the generations. Perceiving and enjoying that smell is made possible by olfactory neurons in the nose, which detect odors and send electrical signals to the brain. The olfactory system is essential for most animals: it supports postnatal survival, detects environmental hazards and mediates emotional, social and nutritional behaviors. It is also one of two systems in the body that can recognize a functionally unlimited number of unknowns, the other being the immune system.
Besides odors, the olfactory epithelium is also particularly susceptible to damage due to viruses, bacteria or toxic chemicals contained within inhaled air. To compensate, it possesses a robust regenerative ability which relies on a resident stem cell population.
Haiqing Zhao is a Professor in the Department of Biology at Hopkins who studies the function and development of olfactory sensory neurons. Recently, he has collaborated with Xin Chen, another Professor in the Department of Biology, and Binbin Ma, a postdoctoral fellow co-mentored by Zhao and Chen, to investigate the role of asymmetric histone inheritance in regulating olfactory stem cell fates during regeneration. Their work has recently been published in Nature Communications.
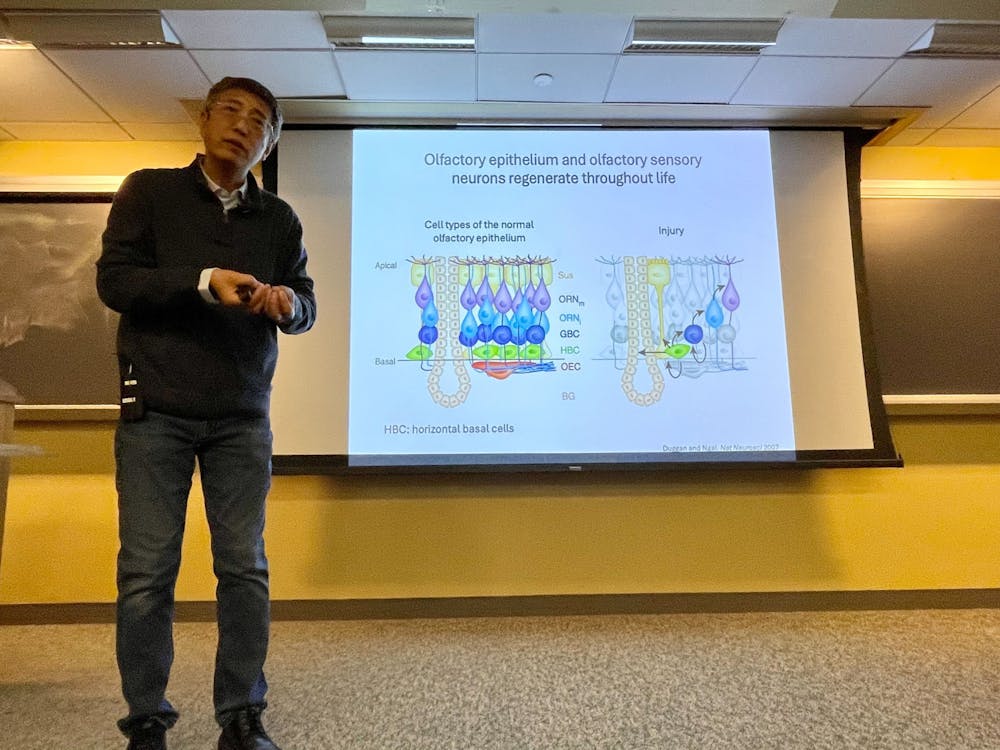
“At the very bottom layer of this [olfactory] epithelium is a layer of cells called horizontal basal cells. These are the bona fide stem cells. These cells allow the tissue to regenerate after injury,” Zhao explained in a March 5 talk to the Department of Biology.
These horizontal basal cells (HBCs) are quiescent under normal conditions but become activated under injury conditions, triggering a cascade of cell proliferation and differentiation that replenishes the epithelium with various cell types, including supporting cells, progenitor cells and olfactory sensory neurons. Over the past two decades, substantial work has been done to study the signaling pathways and receptors by which the activation and differentiation of HBCs are controlled. The Zhao and Chen labs, however, were interested in tackling a different side of the problem.
“How epigenetic mechanisms regulate stem cells — and how they regulate the daughter cells of these stem cells — remains unknown,” Zhao noted. “So now, turning to epigenetics, we come to histones, because histones are genome organizers, and they are also major carriers of epigenetic information.”
The research question was inspired by Chen’s seminal discovery. When a cell divides, the two daughter cells each receive a copy of the genome and also a share of histones that the genomic DNA wraps around. The inheritance of histones, however, may be unequal in certain cell divisions. Asymmetric histone inheritance was first discovered by the Chen lab in 2012 in Drosophila germline stem cells and has since been shown to be essential for Drosophila spermatogenesis.
Due to the observation of asymmetric histone inheritance in invertebrates, Zhao became interested in whether this phenomenon was also present in mammals. The relative histone amount in HBCs could influence how chromatin is organized — and therefore affect how genes are expressed and how cell fate is determined.
However, a few gaps had to be addressed before any experiments on histone inheritance could be done in the HBCs.
“The first thing is that we have to be able to see those cells. Then, we have to make these cells proliferate. Now, we have to be able to see histones in these cells, and we have to be able to catch the very brief moment of this cell dividing,” Zhao said.
Accordingly, Zhao and colleagues used established molecular markers, such as the transcription factor p63 which regulates stem cell fate, to identify HBCs. A single injection of methimazole, a chemical agent which induces widespread cell injury in the epithelium, activates injury responses in the normally quiescent stem cell population. The histones, specifically histone H4, can be tagged with a fluorescent protein called mScarlet in transgenic mice — that is, mice genetically modified to produce this protein.
“About 40% of p63+ cells show this asymmetric distribution of the histones,“ Ma explained. The quantity on one side is more, on the other side is less.”
To better understand this process, the researchers also studied the cells in culture (in vitro), where they could more easily observe and manipulate them.
“Ma observed 48 pairs. So we have 96 cells. This is a really remarkable way — you let them divide, then at this moment [telophase] you physically pick up these two cells and sequence them individually [using single-cell sequencing],” Zhao said.
Even outside the body, HBCs showed similar patterns of asymmetric histone distribution during division. The next step was to investigate the effects of asymmetric histone distribution. At the cellular level, this asymmetry may influence chromatin organization and transcriptional reinitiation, leading to differences in gene expression between daughter cells.
Though the team observed this asymmetric histone inheritance, they were still unaware of whether it had an effect on regeneration. Thus, Zhao wanted to disrupt asymmetric histone distribution to see if there would be any physiological effects on regeneration.
Thus, they adopted a microtubule depolymerizing chemical treatment previously established by the Chen lab to disrupt asymmetric histone distribution.
“All of [the daughter cells] became symmetric… with this treatment, the cells still go to divide and grow, but they all became symmetric,“ Zhao observed.
Returning to a mouse model, the researchers then examined whether disrupting this asymmetry in vivo had functional consequences for tissue regeneration. The drug treatment resulted in less tissue recovery compared to the mock treatment. The behavior of the mice was also examined, especially in regards to food foraging behaviors and odor preference.
“You put the mice in your arena to look for food. You hide the food underneath the bedding. They can only find the food by nose, not by other means. We repeat this many times and count how many of them found the food,“ Zhao explained.
After 15 days, about 75% of control animals were able to find the food, whereas under strong treatment conditions that disrupted histone asymmetry, this dropped to less than 40%. At 28 days, control animals reached near full recovery (about 100%), while treated animals remained at reduced performance at around 80%. Thus, Zhao and colleagues demonstrated that asymmetric histone distribution in HBCs is indeed essential for olfactory stem cell regulation and regeneration.
The next time a familiar smell drifts through the air, it reflects more than just a sensory experience — it is the result of a finely coordinated regenerative system, one that Zhao and his colleagues are helping to decode at the level of stem cells and epigenetics. Their work offers new insight into how the body maintains and restores essential olfactory functions after injury.