An immunotherapy study co-authored in The New England Journal of Medicine by a Hopkins physician offers promising results for those suffering from late-stage melanoma.
Approved by the Food and Drug Administration on March 18, the dual-drug treatment Opdualag can be partially traced back to pharmaceutical company Bristol Myers Squibb. Initial results of the study showed a median survival time without further progression of cancer at 10.1 months, while the drug nivolumab alone led to a median time of 4.6 months.
Dr. Evan Lipson, associate professor of oncology at the School of Medicine, co-authored the study. In total the project encompassed 714 patients and universities in the U.S., Germany, Mexico and beyond. In an interview with The News-Letter, Lipson commented on the motivations behind the study that allowed for the global collaboration.
“We all shared a similar vision. We all cared for these patients with ‘unmet needs.’ We all had a similar focus and knew the rules of the road and how to manage the patients who were receiving these treatments,” Lipson said.
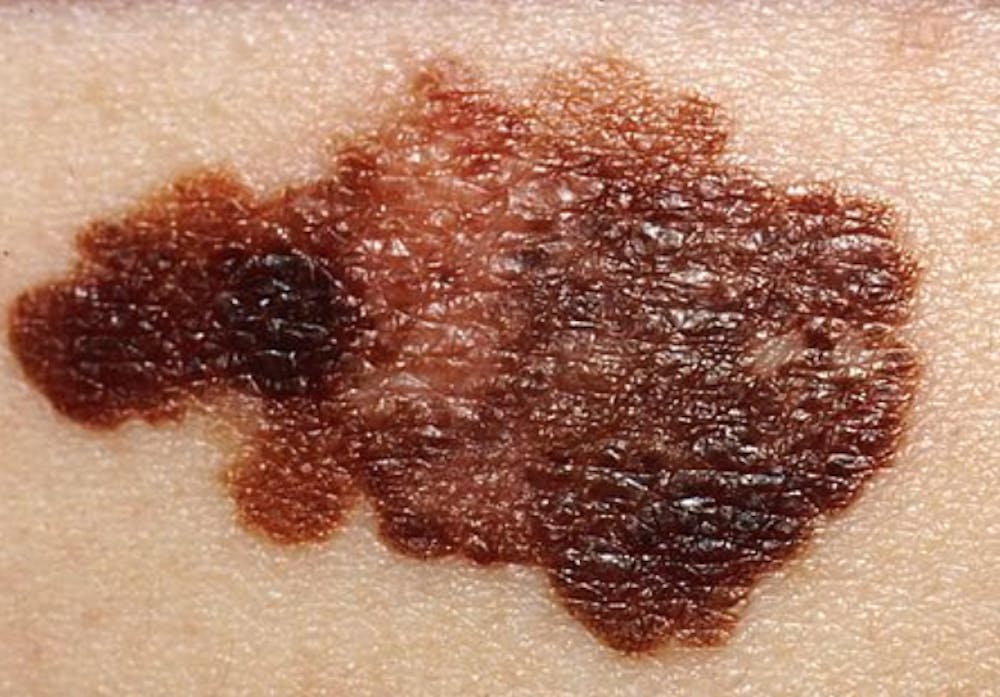
Melanoma is a malignant cancer that is the number-one cause of death among skin cancers despite making up just 1% of all diagnosed skin cancer. It is often characterized by dark splotches on the skin due to the cancer beginning in melanocytes, which are skin cells that control the production of melanin and affect the pigmentation of the skin.
Lipson commented on the difficulties that arise for patients in even recognizing they have melanoma in the first place. Early detection for melanoma is especially important because the rate of survival for localized melanoma is 99%, and for distant melanoma it is just 30%.
“Sometimes the patient isn’t even aware anything is going on. The melanoma could present on their back, which is difficult to see, or underneath their toenail or on the sole of their foot, or in their mouth. We encourage individuals to bring any odd-appearing spots or anything changing, itching or bleeding to the attention of their physicians right away,” Lipson said.
The drugs used in the study were relatlimab and nivolumab. Relatlimab is an antibody known as a LAG-3 inhibitor. LAG-3 is a key immune checkpoint that, when inhibited, can stimulate an immune response against potential cancer cells. Nivolumab has the same function, different only in that it acts on the immune checkpoint known as PD-1.
However, what is stopping scientists from simply releasing large amounts of different checkpoint inhibitors into the bodies of cancer patients? Would this stimulate an aggressive immune response against the cancer?
As Lipson would explain, it might mount an immune response that could do more harm than good.
“The immune system is pretty powerful, so if you get it up and running against the patient’s lung or liver or skin, you can really do some damage,” he said. “The more switches you flip, the more likely you are to wind up with some serious toxicity.”
As for next steps, Lipson and the rest of his team will begin looking into the effects of the combination therapy when used on a more diverse research sample — the study focused exclusively on patients with untreated melanoma. Lipson explained that all of the patients in the study had previously untreated metastatic melanoma, but further research is needed to understand if the treatment will also show such promising effects for those in different stages of melanoma progression.
“For example, for patients who have already had the cancer surgically removed, could we use this therapy to reduce the likelihood of the melanoma ever rising up again? Or can we give them a little bit of this therapy before a surgery?” Lipson said.
Immunotherapy, although a relatively new field, has been seen as the future of cancer treatment. By spurring the body’s immune system to both recognize and attack cancer cells, immunotherapeutic techniques might offer a “universal answer” for cancer.
Lipson offered insights into the ways the field has changed in decades past, explaining how just 15 years prior, there had been little hope for those suffering from distant-stage melanoma. Now, with advancements in modern medicine, the five-year survival rate has jumped to 30%. But as Lipson explained, there is still work to be done.
“We have made huge strides in the last decade-plus. But do I think that a cure for every single patient is around the corner? No, we still have a lot of work to do,” he said. “But are we a whole lot closer? Absolutely.”