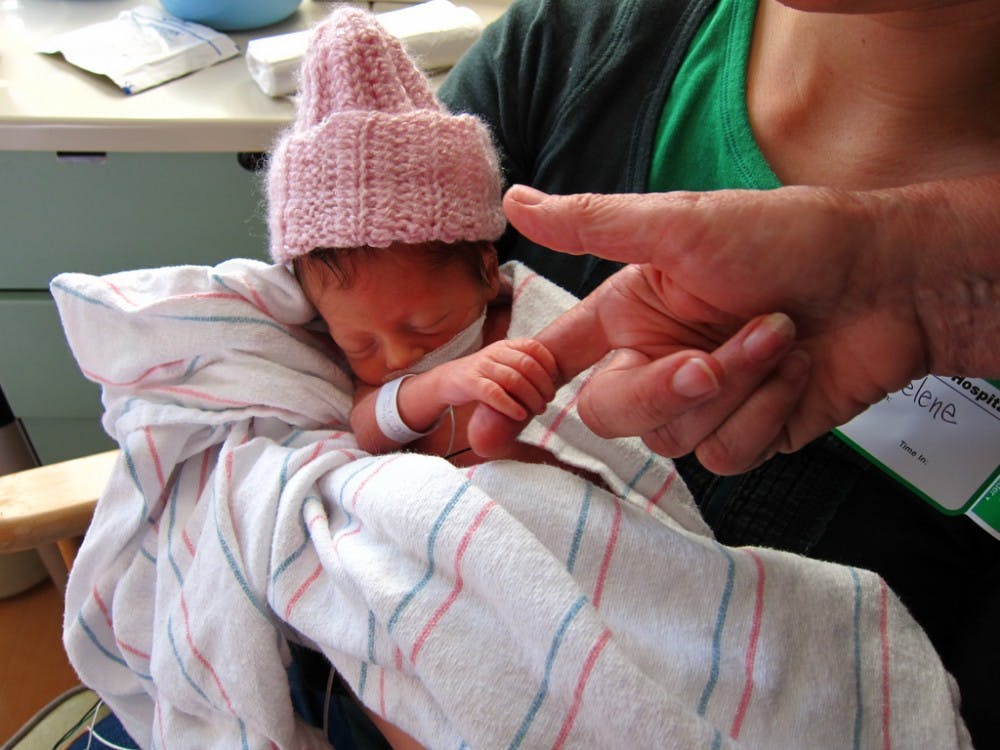
Makena is the only drug to date that has been approved by the U.S. Food and Drug Administration (FDA) for preventing recurrent preterm births. Makena’s chemical compund name is 17-alpha-hydroxyprogesterone caproate (17P) and it was approved by the FDA in 2011.
Since then, the drug was endorsed for widespread use by both the American Congress of Obstetricians and Gynecologists and the Society for Maternal-Fetal Medicine.
More recently, in January 2017, the Society for Maternal-Fetal Medicine repeated its recommendation of 17P in light of the under-utilization of what is deemed an effective drug.
However, a study conducted by researchers at the University of Texas Southwestern Medical Center, has found that the use of 17P had hardly any benefit for expecting mothers with prior preterm births.
Published in the March 2017 issue of the American Journal of Obstetrics and Gynecology, the study details the results of a four year trial from January 2012 to March 2016, during which 430 women with prior preterm births were injected with 17P.
Beginning from 16 to 20 weeks of gestation, pregnant women involved in the study received weekly injections at the Parkland Memorial Hospital in Dallas, Texas.
Having noted the typical rate of 16.8 percent recurrent preterm births at the Parkland Hospital, the researchers evaluated the clinical effectiveness of 17P in reducing the recurrence of premature births.
To compare the premature birth rates, the preterm birth profiles of the study’s participants were matched to similar profiles in the historical cohort.
Based on the mother’s race, body mass index and pregnancy history, 1,290 mothers in the historical control were matched to 430 research participants.
Even after controlling for these demographic factors, 17P did not significantly reduce the rate of recurrent preterm births. In fact, the overall rate of recurrence was higher for the study group treated with 17P at 25 percent.
Researchers observed higher or similar recurrence rates in the 17P study group compared to the historical control for all of the specific pregnancy histories.
For instance, for mothers with one prior preterm birth, the recurrence rate was 31 percent in the 17P study group compared to 18 percent in the historical control.
Moreover, recurrence rates among mothers with three or more preterm births were not statistically different for the 17P group and the historical cohort, which were at 44 percent and 45 percent respectively.
Upon the analysis of blood draws, researchers also discovered that 13.4 percent of the pregnant women treated with 17P were diagnosed with gestational diabetes in contrast to the eight percent of women in the historical control. Researchers found 17P is ineffective at best and at worst, linked to a higher probability of birth complications.
According to the researchers, Makena (17P) was able to receive an accelerated approval from the FDA mostly on the basis of a 2003 study led by Paul J. Meis, a retired professor of obstetrics and gynecology-maternal/fetal medicine at Wake Forest University.
However, questions have been raised following the paper’s publication.
The 2003 study, though completed in two phases, did not include the results of the first phase, which were later reported to have a 36 percent preterm birth recurrence rate in the control group.
On the other hand, the recurrence rate in the control group for the second phase of the study was much higher at 55 percent, leading to speculation that 17P only appeared to be effective because it was compared to an unexpectedly high rate in the control group.
Furthermore, the mechanisms of progestogens, such as 17P, are not well understood. According to the researchers, progestogens were once believed to prevent inflammation in the uterine cervix; however, it was recently discovered that they are not related to reducing the probability of uterine contraction.
On the basis of the 2003 study, pharmaceutical company Lumara Health, formerly KV Pharmaceutical, bought the rights to manufacture 17P and market it under the brand name Makena.
Facing no other competitors, Lumara Health announced the increase of the drug’s cost from $20 to $1,500 per injection in February 2011.
Later that year, Lumara Health lowered the price to $690 per injection, and now, according to Drugs.com, the price is $767.98. In spite of its price gouging, 17P can be found at lower prices ranging from $10 to $25 in the America, albeit in compounded forms at certain pharmacies.
Still, the FDA recommends using the approved Makena except in cases in which the patient has an allergy or medical condition that prevents consumption.
However, as shown by the researchers at the UT Southwestern Medical Center, 17P may not be potent with regard to preventing premature births and may introduce additional complications.
Correction: The drug Makena was originally misspelled as Maketna in an earlier version of this article. The News-Letter regrets this error.












Please note All comments are eligible for publication in The News-Letter.